Between Saturdays: The Things That Age You Faster
From the HRT black box warning the FDA just removed, to what the research says about sunlight and your lifespan, four findings that shift how you think about ageing before you’re old enough.
This edition is about time. Not in an anxiety-inducing way but in the way that the best research on longevity tends to reframe things. The FDA just quietly reversed a 22-year-old warning that has kept millions of women from treatment they needed. A new study on sunlight contradicts several decades of dermatological advice in interesting ways. The gut microbiome is linked to ovarian ageing in ways that will matter for fertility conversations in the next decade. And there’s a finding about menopause and the brain that every woman under 40 should probably know about now.
Caught My Eye…
The FDA Just Removed the HRT Black Box Warning. Here’s What That Means and Why It Was There in the First Place.
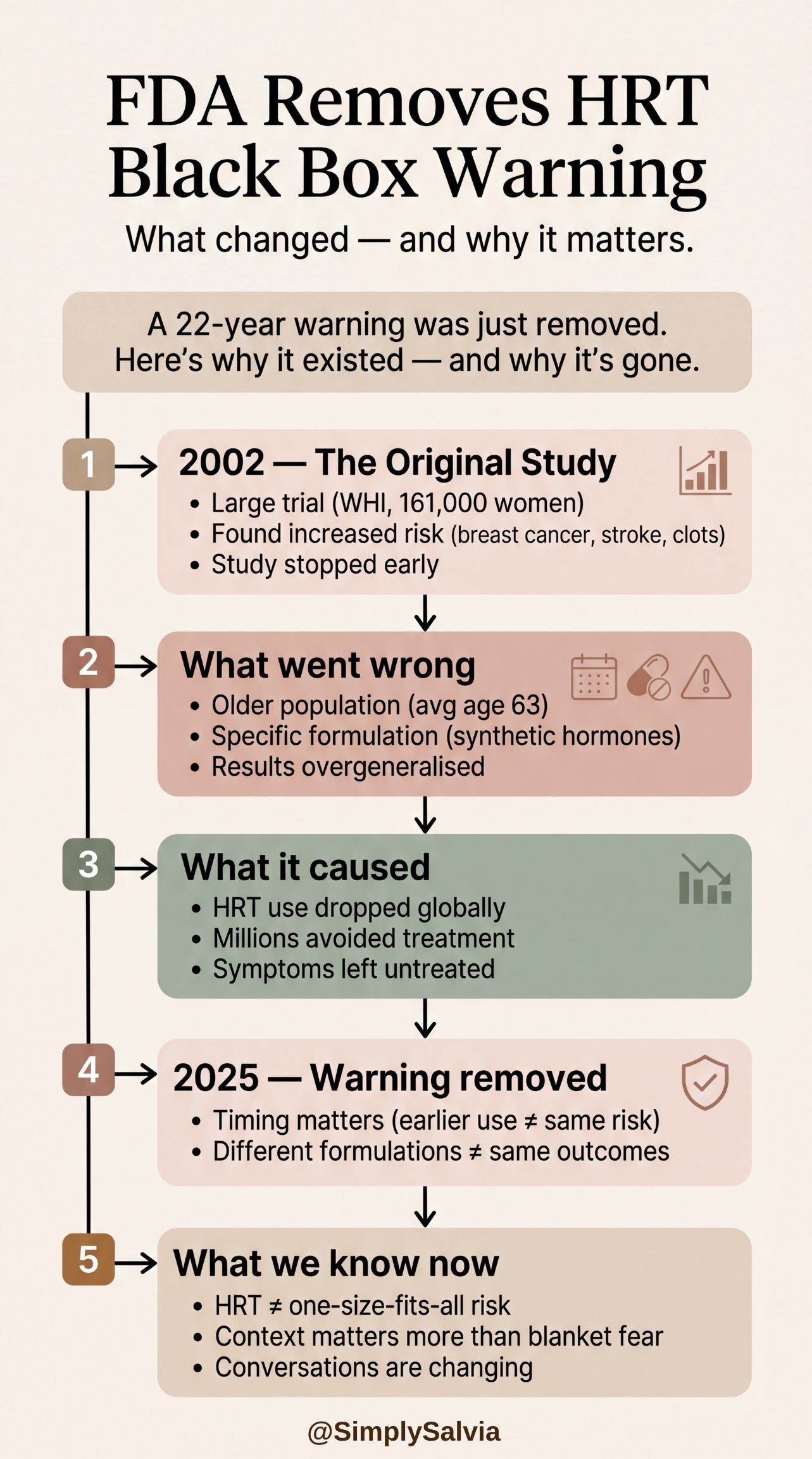
In late 2025, the FDA removed the black box warning (the most serious warning a drug can carry) from menopause hormone therapy products. The warning had been in place for 22 years. FDA Commissioner Marty Makary stated publicly that:
“tens of millions of women have been denied the life-changing and long-term health benefits of hormone therapy because of medical dogma rooted in a distortion of risk.”
This is a significant moment in women’s health. To understand why, you need to understand what put the warning there in the first place.
In 2002, the Women’s Health Initiative, the landmark study of 161,000 women (I’ve discussed in a previous Between Saturdays) published findings from a large trial of combined oestrogen and progestin therapy (the specific formulation was conjugated equine oestrogen plus medroxyprogesterone acetate, which is synthetic progestin). The trial was stopped early after observing higher rates of breast cancer, heart disease, stroke, and blood clots in the hormone therapy group compared to placebo.
The findings were widely reported, widely misunderstood, and dramatically changed prescribing. HRT use plummeted globally. Women who had been on it for years stopped. Women approaching menopause were advised against starting.
What subsequent analysis found: the initial findings were specific to the particular formulation tested (synthetic progestin, not bioidentical progesterone), the age group studied (the average participant was 63, significantly older than the women typically starting HRT for menopausal symptoms), and the duration and dose. Further research found that for women who start hormone therapy in the early menopause transition, within 10 years of menopause and before age 60, the risk profile is substantially different. The cardiovascular risk didn’t replicate. The benefits; relief of vasomotor symptoms, improved sleep, bone protection, and emerging evidence for cognitive protection were real and significant.
The Society for Women’s Health Research documented the consequences of the original interpretation: millions of women undertreated for severely disruptive menopausal symptoms; women aged into significant bone density loss, cardiovascular events, and cognitive changes that earlier intervention might have mitigated.
What this means practically: the black box removal doesn’t mean HRT is risk-free or that it’s appropriate for every woman. Individual medical history, family history, and risk factors all remain relevant. But for women in the early menopausal transition who are experiencing significant symptoms like sleep disruption, hot flashes, mood changes, the conversation with their doctor about hormone therapy is no longer framed by a 22-year-old warning based on a study population that doesn’t represent them.
If you’re in your 20s reading this: this matters to you eventually, and it matters to your mother now. The treatment is evidence-based, the previous caution was based on a misread of a single study, and the reversal is significant.
Menopause Linked to Grey Matter Loss in the Brain. The Implication for Young Women Is About Timing.
A major study published in February 2026 and summarised by ScienceDaily found that menopause is linked to changes in brain structure, mental health, and sleep. Brain scans revealed grey matter loss in areas tied to memory and emotional regulation in menopausal women.
This is part of a larger picture that’s been assembling in the research over the past five years. Oestrogen has neuroprotective properties — it supports neuronal survival, reduces inflammation in brain tissue, and appears to have a role in maintaining the grey matter of specific brain regions. When oestrogen declines during the menopausal transition, those protective effects decline.
The research specifically implicates the hippocampus (memory formation) and regions involved in emotional regulation. Women commonly report “brain fog,” memory lapses, and mood changes during perimenopause and the structural data is beginning to suggest these are not entirely subjective. They may reflect real, measurable changes in brain tissue that occur during the hormonal transition.
The implication for young women is this: the “timing hypothesis” in HRT research suggests that the neuroprotective benefits of oestrogen therapy are greatest when hormone therapy is initiated early in the menopausal transition before significant neurological changes have accumulated. Starting HRT at 65, after a decade of low oestrogen, may not offer the same cognitive protection as starting at 51 when the transition begins.
This doesn’t mean every woman should be on HRT. It means that the conversation about hormone therapy, which previous research had deferred to late life is increasingly being framed as a decision with earlier windows that matter. The oestrogen window for brain protection may be narrower than the window for symptom relief.
It’s also, again, a reminder that perimenopause in women in their 30s is more common than most people know. If you or someone you know is in that age bracket and experiencing unexplained cognitive changes, mood instability, or disrupted sleep, hormones are worth checking.
The Gut Microbiome and Ovarian Ageing: A New Mouse Study That Matters
This is a research finding that’s early-stage and needs the appropriate caveats — it’s in mice, not humans. But the mechanism is so compelling and the implications so significant that it belongs in this edition.
A study published in Nature Aging in March 2026 found that ovarian ageing in female mice is directly linked to gut microbial profiles and that transferring gut microbiota from young mice to older mice produced measurable changes in the ovarian transcriptome, reduced inflammation-related gene expression, and induced transcriptional features the researchers described as consistent with “ovarian rejuvenation.”
In plain language: changing the gut microbiome of older female mice changed what their ovaries were doing, at the gene expression level, in ways that looked more like younger ovaries.
The mechanism the researchers identified involved NAD metabolism and inflammatory pathways in the ovarian tissue. The gut microbiome appears to regulate ovarian function through at least two routes: direct influence on circulating oestrogen levels (through the estrobolome, the set of gut bacteria that metabolise oestrogen), and systemic inflammation pathways that affect ovarian tissue ageing.
Earlier menopause is linked to shorter lifespan and higher risk of cardiovascular disease, osteoporosis, and dementia. If the gut microbiome influences the timing and pace of ovarian ageing and this study provides the most direct evidence yet that it does, then gut health is a fertility and longevity issue, not just a digestive one.
What to do with this: the honest answer is that the specific interventions to translate this into human reproductive health practice don’t exist yet. This is a mechanistic finding in an animal model. But it adds weight to the growing evidence that the gut microbiome is a central regulator of female hormonal health and that the dietary and lifestyle choices that support gut microbiome diversity (high-fibre eating, fermented foods, reduced ultra-processed food, consistent sleep, stress management) are not just gut health choices. They may be, in a meaningful sense, choices about hormonal health, fertility, and the pace of biological ageing.
Sunlight, Vitamin D, and the Nuance the Dermatology Conversation Has Been Missing
For years the public health message on sunlight has been clear and almost one-dimensional: sun exposure causes skin cancer, wear SPF every day, minimise unprotected exposure. This is sound advice as far as it goes. But a growing body of research is adding significant nuance that isn’t reaching most people.
The finding that matters most: vitamin D insufficiency affects approximately 1 billion people globally, with estimates of deficiency particularly high in northern latitudes, among darker-skinned populations (who require more sunlight to produce equivalent vitamin D), and among people who consistently avoid all sun exposure. In the UK, Public Health England estimates that approximately 1 in 5 people have low vitamin D levels.
The health consequences of vitamin D insufficiency are not limited to bone density. Vitamin D receptors are present in almost every cell in the body. Emerging research links insufficiency to higher rates of depression, impaired immune function, increased susceptibility to autoimmune conditions, and disrupted menstrual cycle regularity and higher rates of PCOS symptoms. A 2024 meta-analysis found that vitamin D insufficiency is associated with worse PMS symptoms and more dysmenorrhea, with supplementation producing measurable improvements in both.
The nuance this adds to the SPF conversation: daily SPF on the face is still appropriate and sensible for preventing photoageing and reducing skin cancer risk. But blocking all skin sun exposure entirely, particularly in populations where diet and supplementation don’t fill the gap may be creating a different problem. Brief unprotected sun exposure on the arms or legs (10–15 minutes at midday, 2–3 times per week, outside peak UV months in northern climates) is generally considered sufficient for vitamin D synthesis in lighter-skinned individuals without the cumulative skin damage of prolonged exposure.
If you wear full-coverage SPF daily and eat a typical Western diet with limited fatty fish, eggs, and fortified foods: get your vitamin D levels tested. The target serum level is approximately 50–75 nmol/L; below 30 nmol/L is considered deficient. Winter supplementation of 1,000–2,000 IU daily is widely considered safe and appropriate for populations with limited winter sun.
The evidence suggests the optimal approach is not maximum sun exposure or zero sun exposure. It’s calibrated sun exposure with SPF where it counts (face, décolletage, any chronically sun-exposed area) and adequate vitamin D from some combination of diet, moderate sun, and supplementation.
The information in this post is for educational and informational purposes only. None of the above constitutes medical advice. Always consult a qualified healthcare professional for personal health concerns.
Between Saturdays is a weekly research round-up from Simply Salvia. Four things from science and wellness worth knowing about. If someone sent this to you, you can subscribe here.
Detailed Readings:
Looking Back, Moving Forward: Women’s Health Research in 2025 and the Road Ahead
Estropausal gut microbiota transplant improves measures of ovarian function in adult mice