Between Saturdays: The Science of the Weird Feeling
Four everyday experiences you’ve felt, but never had a name for.
This edition is about the gap between what people experience and what gets studied. The pattern that most women recognise but no one has formally acknowledged. The biological explanation for the feeling that everything is harder in certain weeks. The food habit that affects health in ways you might not expect. And a skin finding that connects something internal — your kidneys, of all things, to something visible on your face.
Caught My Eye…
The Premenstrual Energy Crash Has a Name, a Mechanism, and It’s Not “Just PMS”
Most women are familiar with the pattern: two weeks before your period, something shifts. Energy drops. Motivation drops. Cognitive tasks that felt easy feel effortful. Sleep doesn’t seem to restore the way it usually does. The body feels heavier. This window, roughly days 22 through 28, is usually labelled “PMS” and attributed, with a kind of vague handwave, to hormones.
What the research has been establishing, piece by piece, is a much more specific biological picture that deserves a more specific name and a more specific response.
The mechanism runs through several simultaneous pathways:
The GABA withdrawal pathway. Progesterone’s metabolite allopregnanolone activates GABA-A receptors, the same receptors that benzodiazepines act on. In the late luteal phase, as progesterone falls sharply, allopregnanolone levels fall with it. For women whose nervous systems have upregulated sensitivity to progesterone during the luteal phase, the withdrawal produces a rebound arousal state that looks and feels like anxiety, insomnia, and an inability to wind down. This is the neurochemical reality behind what women describe as “everything feels too loud” or “I’m wired but exhausted.”
The serotonin connection. Oestrogen supports serotonin synthesis. As oestrogen drops in the late luteal phase, the serotonergic buffer on mood and anxiety reduces. The brain is simultaneously losing its primary inhibitory support (GABA via progesterone) and its primary mood-stabilising support (serotonin via oestrogen) in the same 5–7 day window.
The sleep fragmentation. As covered in Deep Dive: Why You’re Always Tired — Even When You Sleep Enough: late luteal sleep is objectively more fragmented, with reduced slow-wave sleep and more nocturnal arousals. The fatigue that compounds through this window isn’t imagined. You’re sleeping lighter and waking more, on a cycle that’s driven by the hormonal withdrawal, not by behaviour.
The blood sugar instability. Progesterone promotes insulin resistance in its dominant phase. As it withdraws rapidly in the late luteal phase, blood sugar regulation can become temporarily unstable. Research has found that women in the late luteal phase have different glucose responses to the same meal than in their follicular phase, and that the 3am waking that’s common in this window is partly driven by nocturnal blood glucose management.
Understanding this combination explains why the typical advice “take ibuprofen for the cramps, it’ll pass,” doesn’t address most of what’s happening. The fatigue, anxiety, insomnia, and cognitive difficulty of the late luteal phase are driven by four specific biological mechanisms that respond to four different interventions:
For the GABA withdrawal: magnesium glycinate in the evening, which activates GABA-A receptors and partially fills the gap left by falling allopregnanolone. Reducing alcohol completely as alcohol compounds GABA withdrawal rather than relieving it.
For the serotonin drop: regular moderate exercise during this phase, which upregulates serotonin synthesis independent of oestrogen. Morning light exposure for its serotonergic effects.
For the sleep fragmentation: earlier bedtime, cooler environment, no high-intensity training in the evening, no alcohol.
For the blood sugar: a protein-and-fat-inclusive dinner, no high-GI snacking in the late evening, consistency in meal timing.
None of this eliminates the late luteal window as a harder stretch. What it does is reduce how much harder it is and shift it from an experience you’re just waiting to survive into one you have a biological map for.
Eating Speed and Metabolic Health: The Research Most People Haven’t Heard
This one is simple, well-established in the research, and almost completely absent from mainstream nutrition conversation.
Eating speed — specifically, eating fast is independently associated with higher rates of metabolic syndrome, obesity, insulin resistance, and type 2 diabetes. Not because fast eaters eat more (though they often do, more on that). But because of a hormonal mechanism that operates on a slower timescale than most people’s meals.
The mechanism: satiety hormones primarily GLP-1, PYY, and leptin are released by the gut in response to food, but they take approximately 15–20 minutes from the start of eating to reach the brain at levels that register as fullness. If a meal is eaten in 7–10 minutes, the satiety signal arrives after the meal is already finished. The brain doesn’t receive the “we’re full” signal until the stomach is already past comfortable capacity.
A landmark Japanese cohort study of more than 59,000 people found that self-reported fast eaters had significantly higher rates of obesity and metabolic syndrome than slow or normal-speed eaters, independent of total calorie intake and after adjustment for confounders. Multiple meta-analyses have since confirmed the association.
For women specifically, there’s a cycle-linked dimension: appetite and eating speed both change across the menstrual cycle. Studies have found that total caloric intake increases in the luteal phase by 100–500 calories per day on average, driven by progesterone and by the body’s increased metabolic demands in the second half of the cycle. Eating speed may also increase in this phase, when cravings are more pronounced and the impulse to eat quickly is higher.
The practical intervention requires no calorie counting, no food elimination, and no expensive supplements. It is simply: slow down. Put the fork or spoon down between bites. Chew fully before swallowing. Aim for a meal that takes at least 15–20 minutes. This gives the satiety signalling system time to do the job it was designed for.
Research on mindful eating specifically has found that slowing eating speed reduces total calorie intake at a meal, improves post-meal satiety ratings, and in longer-term studies reduces BMI and metabolic risk markers. The mechanism is purely physiological, not psychological. The brain needs time to receive the message the gut is sending.
In a food environment designed to encourage fast consumption; grab-and-go packaging, working lunches, screen eating, this is a more subversive intervention than it sounds.
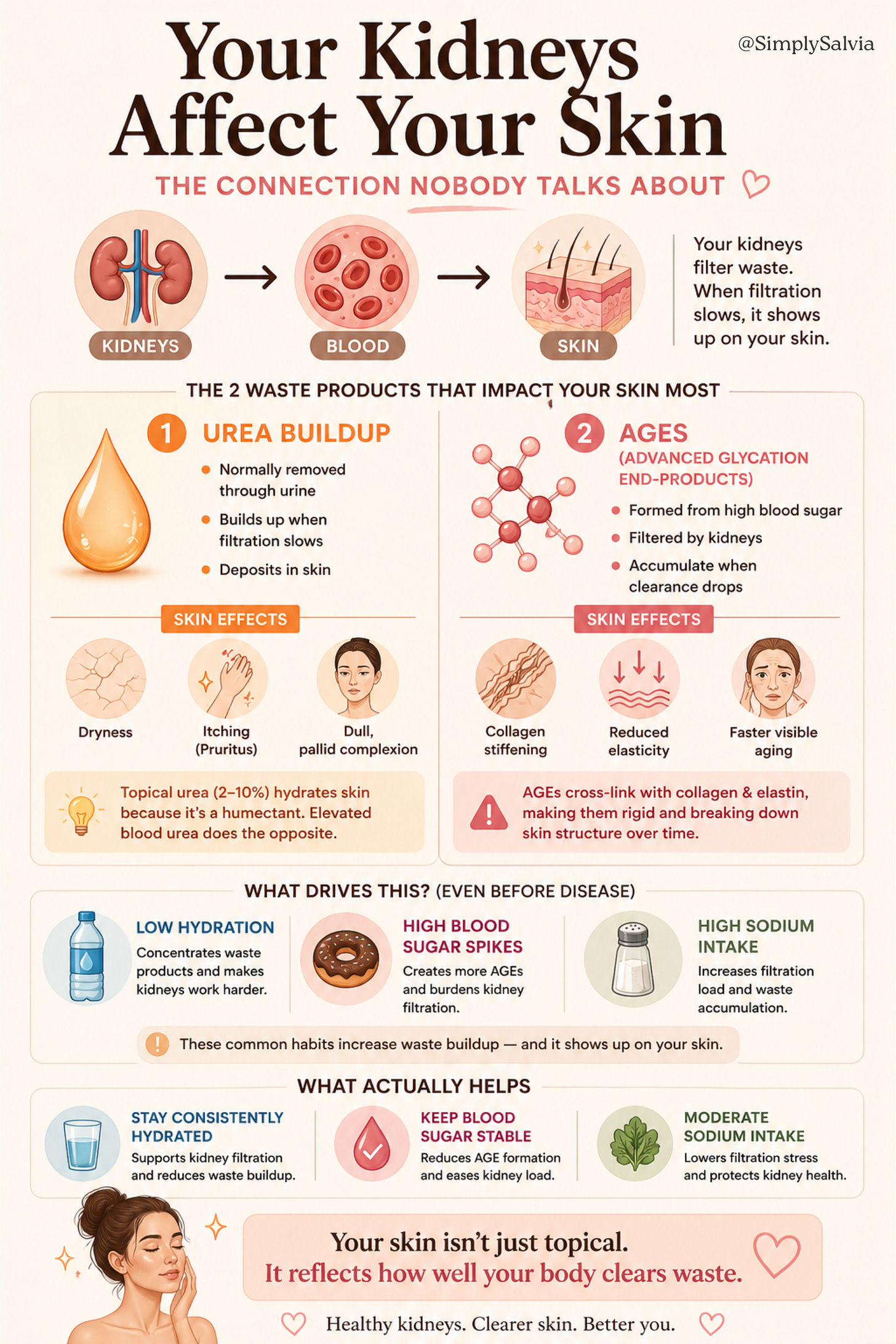
Your Kidneys Affect Your Skin. The Connection Nobody Talks About.
This one surprises people. The skin and the kidneys don’t seem obviously related. But the kidney-skin connection is well-documented in dermatology.
The kidneys’ primary role is filtration: removing waste products, excess fluid, and toxins from the blood. When kidney function is impaired, even subclinically, at levels below what shows up in routine blood tests, waste products accumulate in the bloodstream at levels that have direct effects on skin.
The two most relevant waste products for skin appearance:
Urea. Normally excreted in urine, urea builds up in the blood when kidney filtration is compromised. At elevated blood levels, urea deposits in skin tissue and contributes to dryness, itching (pruritus), and a dull, pallid complexion. Topical urea in low concentrations (2–10%) is actually used as a moisturiser because it’s a natural humectant but systemic urea at elevated blood levels has the opposite effect, disrupting skin barrier function.
Advanced Glycation End-products (AGEs). These are compounds formed when sugars react with proteins, exactly what happens when blood glucose is repeatedly elevated. Kidneys filter AGEs; when kidney function declines, AGE accumulation increases. AGEs directly cross-link with collagen and elastin in skin, making them rigid, reducing their structural function, and accelerating visible ageing. Skin affected by AGE accumulation tends to appear duller, less elastic, and more wrinkled for its biological age.
For a young, healthy person this isn’t about kidney disease. It’s about the factors that affect kidney function subclinically: chronic dehydration (which concentrates waste products), consistently high blood glucose (which accelerates AGE formation and burdens kidney filtration), and high sodium intake (which increases the filtration load). All three are common in typical diets and all three have measurable effects on skin appearance through the kidney pathway.
The practical implications: adequate hydration matters for skin in part because of this mechanism and not because water directly “plumps” skin, which it doesn’t, but because adequate hydration supports kidney filtration efficiency and reduces waste product accumulation. Blood glucose stability matters for skin in part because of AGE formation and not just because of direct collagen glycation, which is the usually-cited mechanism, but because kidneys clearing AGEs adds a secondary buffering system when glucose is controlled.
This is why the skin of people with well-controlled type 1 diabetes and those with poorly-controlled type 1 diabetes looks different, decades before either group develops clinical kidney disease. The metabolic-skin-kidney connection operates at subclinical levels throughout a lifetime.
Women Are More Likely to Attribute Their Own Symptoms to Stress. Research Suggests This Is Medically Dangerous.
This one came out of a finding I kept encountering in different literatures — sleep, cardiovascular health, hormones and it deserves to be named directly.
Research consistently finds that women are more likely than men to attribute their own physical symptoms to psychological causes; stress, anxiety, “overdoing it” and that healthcare providers are more likely to do the same when seeing female patients. This pattern has been documented in cardiovascular medicine (women presenting with heart attacks are more often initially diagnosed with anxiety), in gastroenterology (women with serious GI conditions are more often referred to psychotherapy before organic causes are investigated), and as this newsletter has covered extensively; in sleep medicine, where female OSA, restless legs, and cycle-linked insomnia are routinely attributed to stress or anxiety before a diagnostic workup.
The consequence isn’t just delayed diagnosis. It’s that women learn to interpret their own symptoms through this lens. Persistent fatigue becomes “I need to slow down.” Breathlessness on exertion becomes “I’m out of shape.” Morning headaches become “I’m stressed.” Restless legs become “I’m anxious at night.”
The medical recommendation that emerges from this data, and I want to be direct about it — is: do not accept “it’s probably stress” as the conclusion of an investigation unless investigations have actually been done. Stress is a real and common contributor to many symptoms. It is also a default explanation that, in female patients, has been documented to delay the diagnosis of conditions including autoimmune disease, cardiac conditions, sleep disorders, and hormonal pathologies by years and sometimes decades.
When a symptom is persistent, patterned, and not responding to the stress management interventions that should address it, that is a reason to push for investigation rather than accept the framing. The following is worth knowing: you can ask for specific tests rather than simply describing symptoms. “I’d like my ferritin tested specifically, not just haemoglobin” is a specific request. “I’d like to discuss whether a sleep study is appropriate given these symptoms” is a specific request. Specificity gets results that vagueness doesn’t.
The most important version of this note: heart disease is the leading cause of death in women worldwide. Women are 50% more likely to be misdiagnosed following a heart attack than men, and are more likely to die from a first heart attack. The female presentation of cardiac symptoms; fatigue, jaw pain, nausea, shoulder pain, breathlessness is routinely attributed to anxiety. This is the extreme end of a pattern that operates at all levels of female healthcare.
Know the symptoms. Don’t explain them away before a doctor has investigated them.
The information in this post is for educational and informational purposes only. None of the above constitutes medical advice. Always consult a qualified healthcare professional for personal health concerns.
Between Saturdays is a weekly research round-up from Simply Salvia. Four things from science and wellness worth knowing about. If someone sent this to you, you can subscribe here.
Detailed Readings:
The link between your menstrual cycle and glucose
Self-reported eating rate and metabolic syndrome in Japanese people: cross-sectional study
Dietary energy intake across the menstrual cycle: a narrative review
Mindful Eating: The Art of Presence While You Eat
Women are 50% more likely than men to be given incorrect diagnosis following a heart attack